
Sand-like particles in sludge from landbased salmon farming – origin, significance and implications
Antony Jesu Prabhu Philip, Researcher NOFIMA
FTA Issue No.1 2026
What looks like sand in salmon sludge isn’t sand at all
Sand‑like particles are increasingly observed in sludge from land‑based salmon farming, causing operational challenges and equipment wear. This paper reveals that these particles are not contaminants, but a natural by‑product of the salmon’s own desalination process.

This article can be downloaded as a PDF at the Fjell Tech agenda homepage - Download center.
The potential of salmon farming sludge as a bio-resource is high on the agenda, particularly in Norway. Research initiatives and innovative technologies for collection and utilization of the sludge has received much attention recently.
The composition of the sludge in terms of its nutrient and energy density, as well as levels of minerals, metals and contaminants are the prime factors influencing its utility in downstream processes. In this context, sand-like particles often observed in salmon farming sludges are considered a growing problem causing wear and tear on process equipment used to capture and dewater the sludge. These sand-like particles are reportedly observed when the sludge from land-based farming operations are up-concentrated and collected (Image 1), especially in facilities operating with RAS and high salinity.

Image 1: Up-concentrated sludge from land-based salmon facility showing white, granular crystalline structures. Photo credit: Roy Olav Hovlid.

So far, the origins and implications of these sandlike particles present in the sludge remain largely unknown. Several questions are raised about the origin of these sand-like particles.
Is there sand in salmon feed?
A general misconception is that sand-like particles originate from the feed as impurities in plant derived feed ingredients. On the contrary, the salmon feed does not contain any sand or sand-like particles but indeed contains minerals (ash fraction) such as calcium, phosphorus, magnesium, sodium and potassium, along with essential trace elements such as zinc, copper, manganese, iron and more. The minerals are digested, metabolized and excreted by the fish as dissolved ions via the gills and urine.

Image 2: Author Antony J. P. Philip examining fish to investigate Carbonate precipitates from the gut of Atlantic salmon post-smolt. Photo credit: Thea Morken
A fraction of the minerals are not absorbed by the intestinal system and excreted as particulate matter along with the feces. The minerals originate either from the feed ingredients such as fishmeal which contain hydroxyapatite from the bones of the fish or added to the feed as inorganic mineral salts. The supplemented minerals (inorganic salts) are highly soluble in the gut, whereas the hydroxyapatite is less soluble and has poor digestibility. Hydroxyapatite, although less digestible, does not typically form granular sand-like particles in the feed or fecal material.
The marvelous desalination factory of Salmon
The salmon itself, however, can form crystalline particles during the process of osmoregulation. Osmoregulation is a natural physiological process and a fundamental evolutionary adaptation of life in the aquatic environment. Osmoregulation ensures that the organism maintains the balance between water and ions (salt) in its body in relation to its environment. The salmon begins its life in freshwater rivers (as parr) and moves to the sea (as smolts). The salmon must cope with the change in salinity at sea. The saltwater environment is dehydrating and drives water out of the body through osmosis. Just as we need to drink water to cope with a salty meal, the salmon also needs to drink water to survive. It’s easier said than done.
How it works is a marvel of nature.
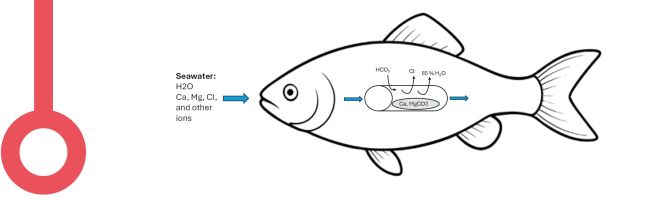
Image 3: Drinking and water uptake mechanism through calcium and magnesium precipitation in the gut of seawater fish.
Drinking is not a challenge, water is aplenty, all the fish must do is to open its mouth. However, the real problem is that the seawater is loaded with salt. The seawater contains mono-valent (Na+, Cl- etc.) and divalent (Ca2+ and Mg2+ etc.) ions. So, the salmon must build its own de-salination plant to produce potable water, which can then be absorbed.
The seawater entering the mouth passes through the esophagus where the monovalent ions are removed (image 3). The seawater along with the divalent ions enters the intestine where they precipitate into calcium-magnesium carbonate (Ca2MgCO3) crystals through secretion of bicarbonate (HCO3-) and increased pH in the intestine (Image 4). The formation of carbonate precipitates reduces osmolality within the intestinal lumen and creates an osmotic gradient for water absorption, i.e. rehydration. These intestinal carbonate precipitates are then excreted from the gut.
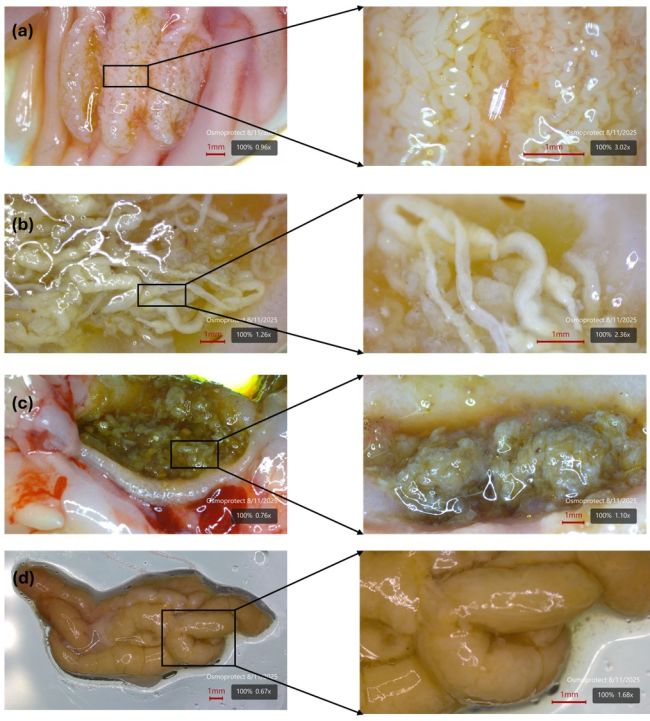
Image 4: The process of carbonate precipitate formation in salmon gut. (a) Dense coils of Ca-Mg carbonates originate in the pyloric caeca; (b) moved to the proximal intestine (c) mixed with digesta and (d) packed into organic mucosal matrix for defecation.

Almost all the fish in the sea have such desalination factory, and it plays a significant role in oceanic CO2 cycling (1). The precipitates are clearly visible when the fish has not been feeding or when their appetite is low, often described as ‘starving feces’ (Image 5). The physical appearance of the Ca2MgCO3 precipitates formed by salmon are ellipsoidal in shape, 2-4 mm in length, appear as white pellets of amorphous crystalline grain-like structures when found intact. They can also be excreted as strings containing many pellets covered with a mucus layer. They comprise of an external organic mucosal matrix covering the inner inorganic mineral structure.
The carbonate precipitates (as is) produced by salmon contain around 30 g/kg calcium, 15 g/kg magnesium, a Ca:Mg ratio of 2:1 (mass ratio) or 1:1 (molar ratio), around 5 mg/kg of zinc and manganese each, and very little phosphorus2 (table 1).
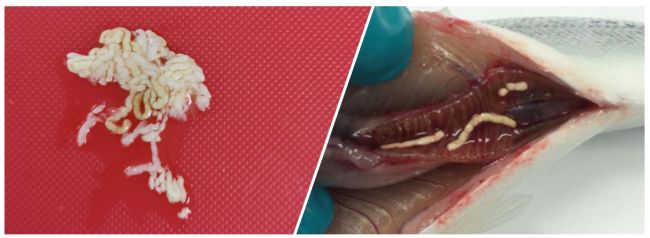
Image 5: Carbonate precipitates from the gut of Atlantic salmon post-smolt after seawater transfer. Photo credit: Antony J Prabhu Philip / Nofima.
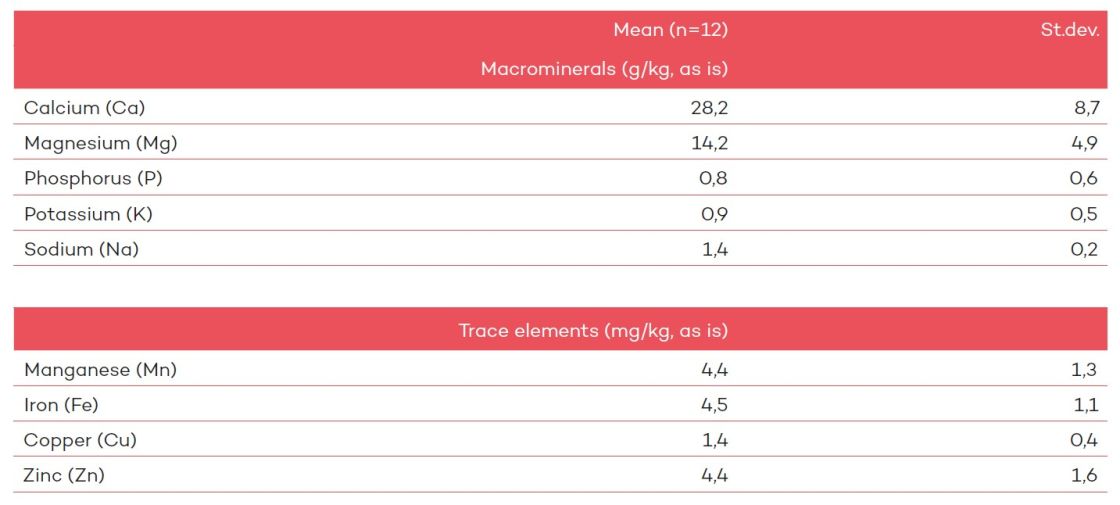
Table 1: Mineral composition of intestinal carbonate precipitates
Can operational procedures at land-based facilities impact the production of sand-like particles?
Land based facilities in Norway have different operational procedures
- Salinity: freshwater, brackish water to saltwater;
- Water temperature: regulated to unregulated;
- Degree of recirculation: flow through systems (FTS) to recirculation aquaculture systems (RAS);
- Smolt-production strategies: photoperiod manipulation, salt feed, seawater addition, combination of one or more;
- Water quality management measures.
Each system contributes differently to the origin and magnitude of these sand-like particles in the sludge / discharge.

Image 6: Atlantic salmon land-based RAS facility. Photo credit: Terje Aamodt / Nofima
The process of drinking and salt precipitation through the gut is a continuous process. As the drinking process is related to hypo-osmoregulatory response, the salinity of water plays an important role in determining the onset and magnitude of drinking, thereby the quantity of precipitates formed.
Drinking in salmon begins and increases gradually until 11-12 ppt (the isosmotic point of salmon), and rapidly beyond 15 ppt3. In full strength seawater, a 100 g salmon drinks seawater to the magnitude of 5-15% of its body weight and produces between 15 to 30 mg of carbonate precipitates every day, depending on water temperature and feed composition.
Change in water temperature can alter appetite and drinking rate thereby affecting the amount and proportion of these precipitates in the sludge/discharge. Smolt feed formulations with different electrolyte (ion) balance can also induce or reduce carbonate precipitate formation.
The proportion of the sand-like granules in the sludge may also vary during the production cycle, for example it can be higher during the smolt-window when a hypo-osmotic state is developing, and appetite is low, or in large post-smolts maintained in brackish water around and above isosmotic point (12 ppt).

Image 7. Wet sludge from wastewater and sludge treatment. Photo credit: Terje Aamodt, Nofima.
Implications for sludge handling and management – A technology & sustainability perspective
The appearance and up-concentration of these sandlike mineral carbonates in the sludge has its implications. While the production of carbonate precipitates by salmon is a fundamental physiological response, bicarbonate salts used for water quality management in saltwater RAS systems can also lead to operational challenges by the precipitation of Ca and Mg salts in the discharge.
Mineral carbonate precipitates of fish origin are macromolecular granules that can clog particle filtration systems (eg. drum filter) leading to increased energy use and lower efficiency. As of today, it is primarily an issue for land-based operations but can potentially be encountered in different forms of closed-containment aquaculture systems (CCS) emerging in Norwegian salmon farming.

The precipitates can enrich the mineral concentration (primarily, Ca and Mg) of the sludge for further sustainable downstream applications like manure / feed material for insects, polychaetas etc. Downstream bioreactor processing of the sludge (e.g. biogas production) can release the bound CO2 leading to increased emissions.
The ‘sand-like particles’ will be part of the salmon sludge. Technologies and applications for industrial scale collection, handling or utilization of sludge from salmon farming should consider these physiological variables. Technological advancements in sludge management should therefore not happen in isolation, but in harmony with salmon biology to advance this field. Understanding salmon biology is central for advancing salmon farming, including sludge and effluent management.
References
1. R. W. Wilson et al., Contribution of Fish to the Marine Inorganic Carbon Cycle.Science323,359-362(2009). DOI:10.1126/science.1157972
2. Philip, Antony J. Prabhu, et al. “Dietary Electrolyte Balance of Atlantic Salmon (Salmo Salar) Freshwater Feeds: Impact on Osmoregulation, Mineral Metabolism and Performance in Seawater.” Aquaculture 546 (January 2022): 737305. https://doi.org/10.1016/j.aquaculture.2021.737305.
3. Ciavoni, Elisa, Johan W. Schrama, Gopika Radhakrishnan, Øystein Sæle, and Antony J. Prabhu Philip. “Salinity Induced Changes in the Progression of Water, Ion and Nutrient Fluxes along the Gastrointestinal Tract of Atlantic Salmon Smolt (Salmo Salar).”Aquaculture 580 (February 2024): 740331. https://doi.org/10.1016/j.aquaculture.2023.740331.
Fjell Tech Agenda is a publication by Fjell Technology Group aimed at sharing knowledge with customers, partners, and stakeholders. Its purpose is to present complex topics in a clear and accessible format for non-experts. The publication is not intended to promote Fjell Technology Group’s products or services. All content reflects the views and insights of the individual author, and Fjell Technology Group does not assume responsibility for the opinions expressed.
Suggestions for future topics are welcome at post@fjelltg.com.
Happy reading!
Antony Jesu Prabhu Philip, PhD, Senior Researcher at Nofima
Antony Jesu Prabhu Philip specializes in minerals and nutritional physiology of fish to understand how physiological and environmental adaptations are challenged by modern aquaculture practices, particularly in salmon farming. He has a dual PhD (Wageningen University, Netherlands & AgroParisTech/ INRAE, France) in aquaculture nutrition with a medal of excellence (Médaille d’Argent Dufrenoy de l’Académie) from The French Academy of Agriculture. for his work on minerals in fish.

